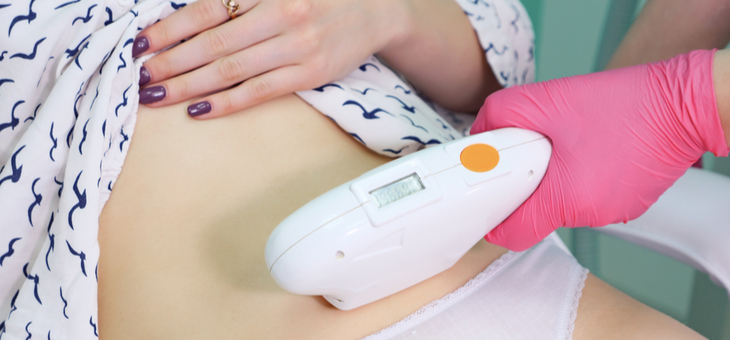
Trials show that a laser light therapy can improve the lives of Parkinson’s sufferers.
Australian med-tech company SYMBYX, in collaboration with Parkinson’s South Australia (PSA), has been developing a laser light therapy called photobiomodulation (PBM) aimed at treating the symptoms, with very promising results.
Treatment involves patients using a hand-held laser unit that emits a non-heating light beam. The beam is directed at the patient’s gut, which is the source of most of the body’s dopamine, a chemical messenger that is necessary for smooth, steady body movements. The high-frequency light stimulates the gut and promotes dopamine production.
The team has published the results of a 52-week trial, involving two groups of patients, that showed improvements across a range of motor and non-motor skills.
More than one million Australians are living with Parkinson’s disease and the condition affects one in 100 people aged 60 and over. It cannot be cured, but the symptoms can be managed.
Read: Billy Connolly: I don’t want to be on stage with Parkinson’s
Symptoms can include muscle rigidity, tremor, difficulty walking, depression, anxiety, difficulty swallowing and loss of sense of smell.
Parkinson’s is classified as a progressive neurological condition and primarily affects a person’s ability to control their body movements. The symptoms are highly individualised, and no two people suffering with Parkinson’s will experience the same thing.
The effects are caused by the degradation of nerve cells in the middle part of the brain, which causes a drop in dopamine.
The SYMBYX trial was conducted in Adelaide and involved two groups of six patients who had been diagnosed with Parkinson’s. Participants used the laser light device in a clinical setting for 12 weeks, and then for a further 40 weeks at home.
Read: Get fit to fight Parkinson’s
The research team found measures of mobility, cognition, dynamic balance and fine motor skill were significantly improved with the PBM treatment. Importantly, they also found no evidence of any adverse side-effects.
“Many individual improvements were above the minimal clinically important difference – the threshold judged to be meaningful for participants,” the study says.
“Individual improvements varied but many continued for up to one year with sustained home treatment. No side-effects of the treatment were observed.”
The researchers noted that the improvements were maintained as long as the patient continued the treatment.
Read: Drinks a neuroscientist recommends for better brain health
The results show that a home therapy administered by patients themselves is possible. Although this first trial is small, it’s the precursor to a larger trial set to begin this month at the Sydney Adventist Hospital.
“Parkinson’s disease is now the world’s fastest growing neurological disease,” says SYMBYX chief executive Dr Wayne Markman.
“This is a promising trial that provides a clear impetus for additional higher-level trials.”
Do you have a family history of Parkinson’s disease? Do you think this treatment could help you or someone you know? Let us know in the comments section below.
If you enjoy our content, don’t keep it to yourself. Share our free eNews with your friends and encourage them to sign up.